Abstract: Natural killer cells(NK cells) are important effector cells in innate immune system, and their roles in anti-virus and anti-tumor immunity are also significant. High purity NK cells are usually separated from peripheral blood mononuclear cell via density gradient centrifugation and magnetic-activated cell sorting(MACS). Cell amplification in vitro via IL-2 or IL-15 maintains activity and function. Cultured cell recognition can be conducted via flow cytometry. Typical phenotype is CD3⁻CD56⁺. Analysis with other markers can evaluate the purity, subpopulation and activation state. The technical system above strongly supports fundamental research and clinical transformation.
Keywords: NK Cell Recognition, Flow Cytometry, Cell Culture, Mass Cytometry
1. NK Cell Culture
1.1. Materials and Devices
The experiment is involved in separation and culture of NK and NKT cell, stimulated and amplified by cytokines(e.g. rhIL-2, rhIL-18 and rhIL-21). Growth conditions are optimized with serum-free medium(PRIME-XV NK Cell CDM). Key devices and consumables include CO2 incubator, centrifuge, cell counter, MACS system, flow cytometer, pipette and centrifuge tube etc, ensuring accuracy and reproducibility of cell operation.
1.2. Cell Culture Medium Preparation
Preparation for NK cell culture medium(PRIME-XV NK Cell CDM): Prepare 1x inoculation medium with rhIL-2(500 IU/mL), rhIL-2(10 ng/mL), rhIL-18(10 ng/mL) and rhIL-21(10 ng/mL) respectively, and 2x expansion medium with doubled concentration. Before use, preheat the culture medium to 37℃. Frozen or fresh cells are thawed or transferred with 15mL centrifuge tube containing 10mL preheated culture medium. Centrifuge at 300g for 5 minutes. Discard supernatant and resuspend. PBMC enriches NK cell via magnetic beads or flow cytometry sorting, using CD56 positive or CD3 negative selection. After sorting, conduct cell counting. Then, detect cell viability and CD3⁻CD56⁺ phenotype via flow cytometry, and evaluate enriched effects.
1.3. NK Cell Culture
Resuspend enriched NK cell in 1x inoculation medium containing cytokines at 0.5x10⁶ cells/mL. Add activated magnetic beads for stimulation. Inoculate 2mL cell suspension into T-25 flask, and culture at 37℃ for a maximum of 14 days with 5% CO2 under humidified condition. Supplement equivalent volume of 2x expansion medium every 2-3 days to maintain cytokine concentration. If necessary, transfer to a larger container to increase culture volume. Centrifugation is not required, but regular detection of cell density, viability and CD3⁻CD56⁺ phenotype monitors expansion effects and cell state.
1.4. Preparation of NKT Cell
Before use, preheat PRIME-XV NK Cell CDM to 37℃ for 15min at least. Avoid repeatable heating and cooling. Frozen or fresh cells are transferred to 15mL centrifuge tube containing 10mL preheated culture medium. Centrifuge at 300g for 5 minutes. Discard supernatant and resuspend cell deposits with less liquid. PBMC enriches NKT cell via magnetic beads or flow cytometry sorting, analyzing cell counting and phenotype.
1.5. NKT Cell Culture
Resuspend NKT cell in PRIME-XV NK Cell CDM with IL-2(1000 IU/mL). Adjust density at 0.5×10⁶ cells/mL. Inoculate in T-25 flask at 2 ml/bottle. Vertically culture cell in humidified incubator for a maximum of 14 days with 5% CO2. Supplement 2x expansion medium every 2-3 days to double volume. Use a larger container, if necessary. Centrifugation is not required, but regular detection of cell density, viability and phenotype monitors expansion state.
2. Flow Cytometry Analysis
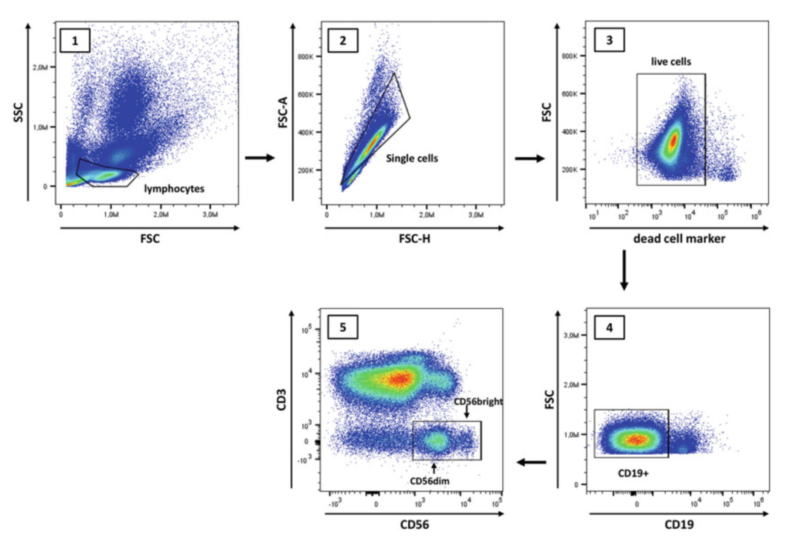
Prepare single cell suspension(e.g. whole blood, PBMC or tissue samples) and adjust cell density at 2x10⁵/tube. Using fixable viability dye can wash after staining. Otherwise, add nucleic acid viability dye after surface staining. Block Fc first. Then, add surface antibodies(e.g. CD45, CD3, CD56, CD16 etc) and incubate for 20min at 4℃ in the dark. Detection of intracellular markers adds intracellular antibodies after fixation and permeabilization. Incubate on ice in the dark for 40-60min. Wash and resuspend in DPBS. Keep the sample at 4℃ and conduct the assay timely in the dark. Before the experiment, control setting of blank, single staining and fully stained tubes can ensure accurate results.
3. Mass Cytometry
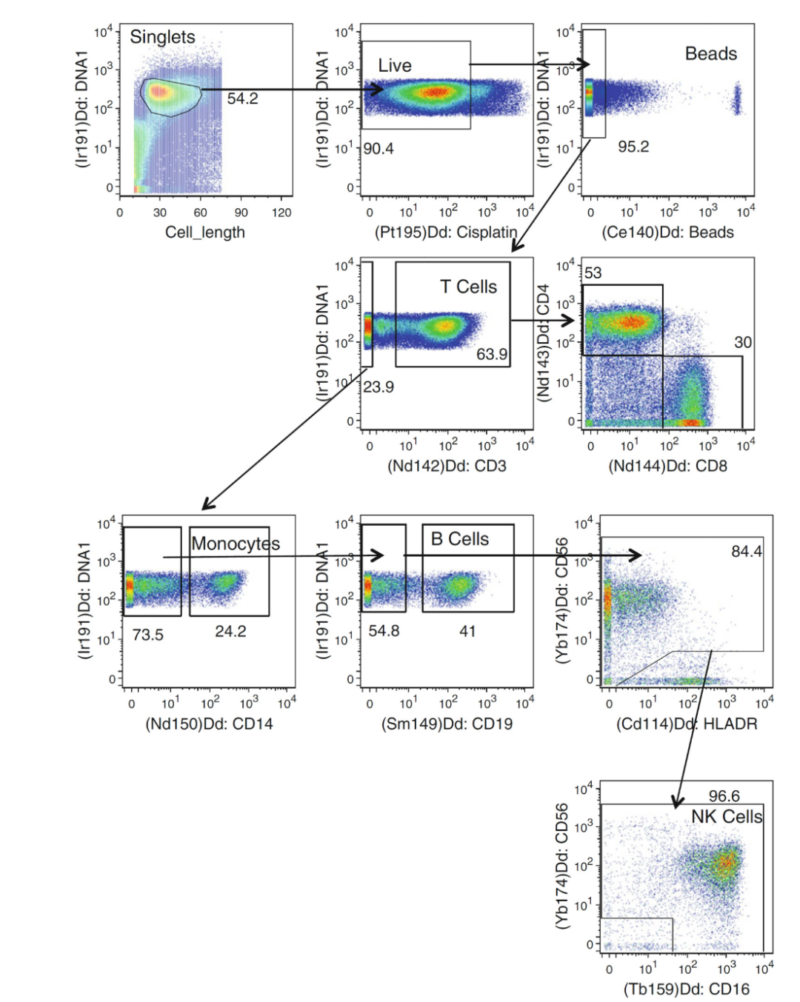
Compared with flow cytometer, CyTOF can detect over 40 parameters at single-cell level using heavy metal isotope labeled antibodies. The principle of accurate cell recognition for phenotype depends on signal detection of rare-earth metal with different mass number, e.g. NK cell subpopulation. Overlap-free metal signal doesn't require for fluorescence compensation and improve data accuracy. The technology avoids spectral interference and greatly extends multicolour analysis, strongly supporting high-dimensional immunophenotyping.
4. Significance of NK Cell in Immunological Research
NK cell can be separated from peripheral blood via density gradient centrifugation and magnetic-activated cell sorting(MACS), also cultured via cytokines(e.g. IL-2 etc) induced in vitro amplification. Cultured cells are identified via flow cytometry. The typical phenotype is CD3⁻CD56⁺, analyzing purity and subpopulation via markers like CD16 etc. This method supports NK cell function research and clinical applications, and also plays a key role in immunological research.
| Recommended Products | |||
| Species | Cell Populations | Flow Cytometry Antibody Combination | Cat.No |
| Human | T/B/NK cell populations detection | CD45-PerCP | PCP-30039 |
| CD3-FITC | FITC-30004 | ||
| CD16-PE | PE-30061 | ||
| CD56-PE | PE-30008 | ||
| CD19-APC | APC-30066 | ||
| Human | Thl/Th2 cell populations detection | CD3-PerCP/Cyanine5.5 | PCP55-30004 |
| CD4-FITC | FITC-30005 | ||
| IFN-γ-PE | PE-30053 | ||
| IL4-APC | APC-30043 | ||
| Mouse | Thl/Th2 cell populations detection | CD3-PerCP/Cyanine5.5 | PCP55-30002 |
| CD4-FITC | FITC-30128 | ||
| IFN-γ-PE | PE-30074 | ||
| IL4-APC | APC-30026 | ||
| Human | Treg cell populations detection | CD4-FITC | FITC-30005 |
| CD25-PE | PE-30035 | ||
| CD3-PerCP-Cy5.5 | PCP55-30004 | ||
| CD127-FineTest®647 | F647-30033 | ||
| Mouse | Treg cell populations detection | CD4-FITC | FITC-30128 |
| CD25-APC | APC-30017 | ||
| FOXP3-PE | PE-30111 | ||
REFERENCES
[1]Suppression of LKB1-mutant lung adenocarcinoma by natural killer cells from females, PMID: 40511603.
[2]cGAS/STING-mediated upregulation of NKG2D ligands in LSCs contributes to enhanced sensitivity to NK cells, PMID: 41809821.
[3]Transcriptome sequencing of Hodgkin lymphoma Hodgkin and Reed-Sternberg cells reveals escape from NK cell recognition and an unfolded protein response, PMID: 40799573.
