Abstract: Flow cytometry detects cell surface markers(e.g. CD molecules) via fluorescence-labeled antibodies, and is widely applied in typing and functional analysis of immune cells. Besides surface flow cytometry staining, quantitative analysis can be performed for intracellular or nucleus cytokines, functional proteins and DNA content using fixation and permeabilization technology. Membrane permeability of different dyes is also varied. Processing methods depend on localization of target antigen. General analysis for cellular phenotype, activation state, proliferation and apoptosis can use various technologies like multi-color labeling, intracellular staining and nucleic acid dyes, technically supporting immunological research and clinical diagnosis.
Keywords: Flow Cytometry Staining, Antibody Immunostaining, Dump Gating, Intracellular Staining, Dye Staining
1. Antibody Immunostaining
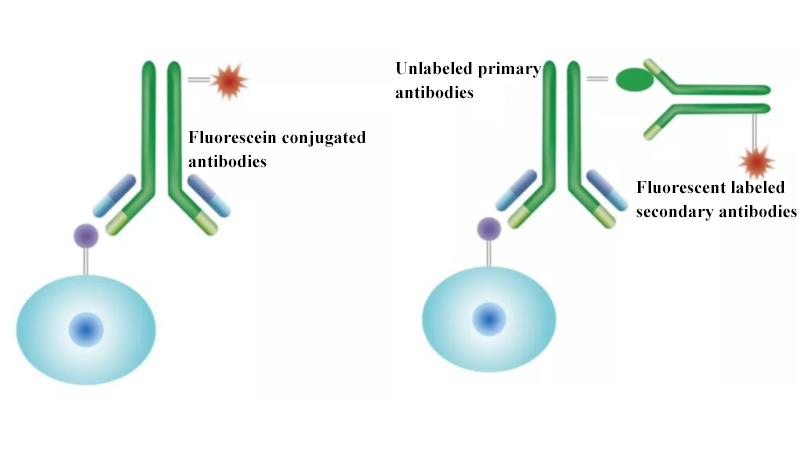
Direct and indirect immunostaining are commonly used in flow cytometry. In direct immunostaining, fluorescein conjugated antibodies bind with cell surface antigen. After washing, detection of fluorescence signal is easy and rapid. In indirect immunostaining, specifically unlabeled primary antibodies are first added to bind with the target antigen. After washing off unbound antibodies, fluorescent labeled secondary antibodies are added to recognize primary antibodies and form antigen-antibody-secondary antibody complex. Fluorescence signals are amplified. This method improves detection sensitivity via signal amplification effects of secondary antibodies, and is suitable for detecting lowly expressed antigen. Complex steps indicate higher non-specific risk.
2. Dump Gating
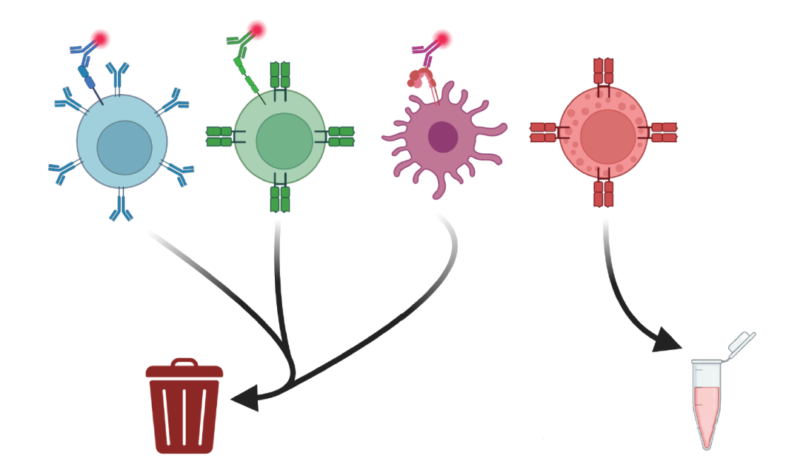
During processing heterogeneous cell mixture, negative selection is suitable for sorting specific cell populations(e.g. CD8⁺T cell) and avoiding antibody interference. Traditional MACS method is based on sorting single marker. FACS can synchronously use multiple non-lineage markers to achieve higher purity of sorting. Dump gating is highly effective. All positive signals are removed via the same fluorescence-labeled lineage markers(e.g. CD4, CD19, CD11c) of non-target cells(e.g. CD4+ T, B cell and dendritic cell). Only negative population is retained. Enrichment of unlabeled target cells reduces non-specific binding, and also improves sorting accuracy and compatibility of downstream applications.
3. FACS Intracellular Staining
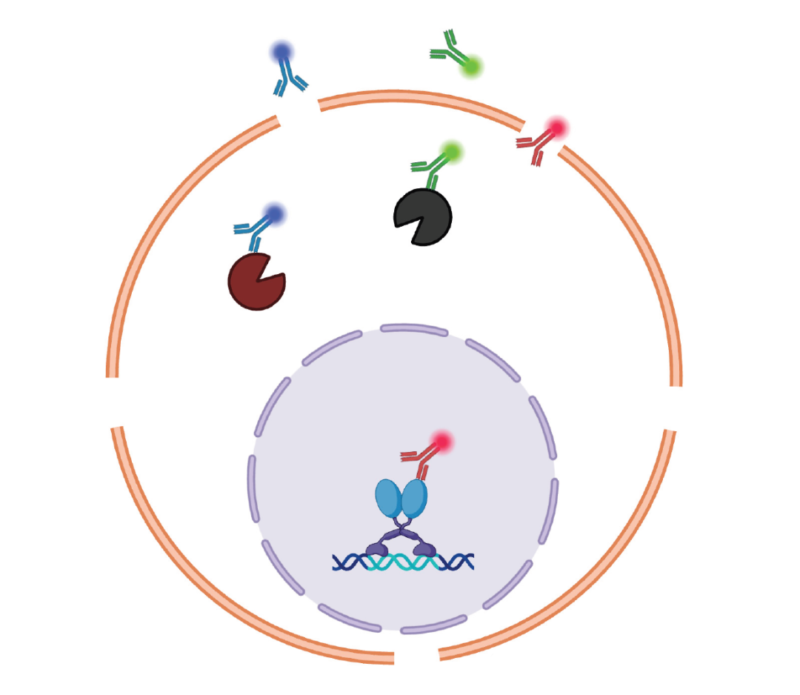
Antibodies enter target proteins in the cell via fixation and permeabilization. Fixation can cross-link proteins and stabilize cell structure. Permeabilization forms pores on the membrane, facilitating antibody permeability. This process happens after surface and activity staining. Cytoplasmic and nuclear proteins(e.g. cytokines, kinase, transcription factors, histones etc) require for different fixation/permeabilization buffer kits. Labeling reagents in the nuclear can synchronously detect cytoplasmic and nuclear proteins. Besides, samples processed via fixation/permeabilization can be temporarily stored at room temperature, improving experimental flexibility.
4. Dye Staining
Dye staining is the non-antibody dependent flow cytometry assay via binding with DNA or free amino group on the protein. Common applications include cell proliferation and activity analysis for living cells. Esterification dyes can unconventionally enter cells, transformed into fluorescence and membrane-impermeable products via enzymolysis. Fluorescence intensity gradually decreases with cell division, tracking proliferation situation. Nucleoside analogues like BrdU can be mixed into newly synthesized DNA. Cellular viability dyes are impermeable, and only enter membrane-damaged dead cells. Binding with intracellular free amine group produces strong signal. Living cells can be hardly stained, facilitating distinction and selection of living cell population during analysis.
Indirect labeling can be used when conjugated antibodies are unavailable. Biotin-streptavidin or host reaction amplifies signal. Dump gating labels non-target cells via the same color, efficiently sorting target cells with clean surface. Detection of intracellular targets requires for fixation and permeabilization after surface staining, distinguishing cytoplasmic or nuclear markers. Besides, small molecular dyes can bind with DNA or free amine group, and are applied in proliferation of living cells and activity analysis. Flexible applications are featured with membrane permeability and impermeability.
| Recommended Products | |||
| Species | Cell Populations | Flow Cytometry Antibody Combination | Cat.No |
| Human | T/B/NK cell populations detection | CD45-PerCP | PCP-30039 |
| CD3-FITC | FITC-30004 | ||
| CD16-PE | PE-30061 | ||
| CD56-PE | PE-30008 | ||
| CD19-APC | APC-30066 | ||
| Human | Thl/Th2 cell populations detection | CD3-PerCP/Cyanine5.5 | PCP55-30004 |
| CD4-FITC | FITC-30005 | ||
| IFN-γ-PE | PE-30053 | ||
| IL4-APC | APC-30043 | ||
| Mouse | Thl/Th2 cell populations detection | CD3-PerCP/Cyanine5.5 | PCP55-30002 |
| CD4-FITC | FITC-30128 | ||
| IFN-γ-PE | PE-30074 | ||
| IL4-APC | APC-30026 | ||
| Human | Treg cell populations detection | CD4-FITC | FITC-30005 |
| CD25-PE | PE-30035 | ||
| CD3-PerCP-Cy5.5 | PCP55-30004 | ||
| CD127-FineTest®647 | F647-30033 | ||
| Mouse | Treg cell populations detection | CD4-FITC | FITC-30128 |
| CD25-APC | APC-30017 | ||
| FOXP3-PE | PE-30111 | ||
REFERENCES
[1]Staining Triad: A fully automated and zero-waste flow cytometry staining system fostering the 3R to 4R transition, PMID: 40882705.
[2]Measuring Chimeric Antigen Receptor T Cells (CAR T Cells) Activation by Coupling Intracellular Cytokine Staining with Flow Cytometry, PMID: 41478983.
