Product Name:2019-nCoV IgM/IgG Antibody Detection Kit (Colloidal Gold)
Sample: Plasma, Serum
Reactivity: Human
Product Description
This kit is used to qualitatively detect 2019-nCov IgM/IgG antibody in human serum & plasma sample in vitro. It only can be used as a supplementary testing of 2019-nCov in suspected cases. It can’t be used to make a definite diagnosis of 2019-nCov disease, still needs to work together with nucleic acid testing. Product is for scientific institution use only.
Principle of Detection
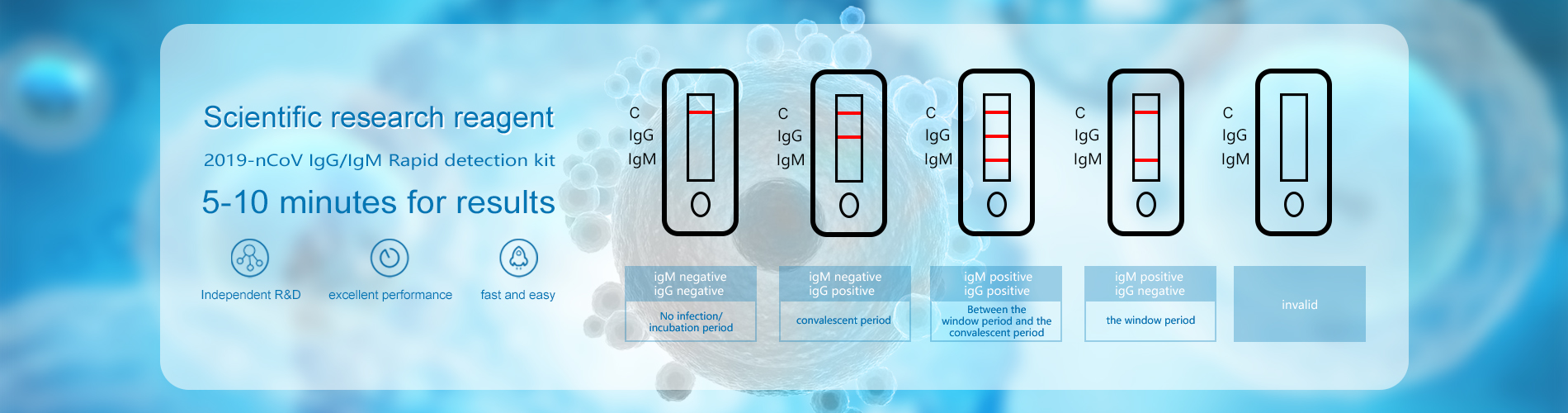
The test is performed using capture method by solid phase immunochromatography. The test sample (blood/plasma) diffuses upward by capillary force, when passes through the marker pad, the 2019-nCov IgM/IgG antibody will react with the 2019-nCoV antigen colloidal gold complex. The new formed colloidal gold-labeled antigen-IgM complex and colloidal gold-labeled antigen IgG complex will diffuse onto the nitrocellulose membrane together with sample, then react with the mouse anti-human IgM antibody which is coated on the T1 line (detection line), then T1 line turns red. The non-react colloidal gold immune complex continues to move up and is blocked by T2 line (detection line) which coated with the mouse anti-human IgG antibody, then T2 line turns red. Finally, the remaining un-intercepted colloidal gold conjugates continued to move up and combine with the C line (quality control line), indicating that the whole process is finished.
Product Components
| Components | Ingredient |
| Detection Card | Foil bag, Desiccant, Test Strip and plastic card.
Test Strip is composed of absorbent paper, cellulose nitrate membrane, sample pad, colloidal gold marker pad and PVC plate. 1.0 mg/ml mouse anti-human IgM antibody has been coated on T1 line 1.0 mg/ml mouse anti-human IgG antibody has been coated on T2 line 1.0 mg/ml internal reference protein c has been coated on C line The marker pad contain 40 OD recombinant 2019-nCov antigen Colloidal Gold complex |
| Sample dilution | HEPES buffer containing Casein (0.1M), 5 ml/bottle |
| Dropper | 50 pieces/kit |
P.S.: You can’t mix components from different batches.
