FineTest Elisa kit contributes to the research on ovarian cancer and nanomedicine. The immunoassay is designed to measure PRKCSH(glucosidase 2 subunit beta) concentration in plasma.
Publication Details
Article Title: Nano-scavengers for Blood Biomarker Discovery in Ovarian Carcinoma
Journal Title: Nanotoday
DOI: 10.1016/j.nantod.2020.100901
IF: 20.722
Abstract: The development and implementation of biomarker-based screening tools for ovarian cancer require novel analytical platforms to enable the discovery of biomarker panels that will overcome the limitations associated with the clinically used CA-125.The systematic discovery of protein biomarkers directly from human plasma using proteomics remains extremely challenging, due to the wide concentration range of plasma proteins. Here, we describe the use of lipid-based nanoparticles (NPs) as an ‘omics’ enrichment tool to amplify cancer signals in the blood and to uncover disease specific signatures. We aimed to exploit the spontaneous interaction of clinically-used liposomes (Caelyx®) with plasma proteins, also known as’ protein corona’ formation, in order to facilitate the discovery of previously unreported differentially abundant molecules. Caelyx® liposomes were incubated with plasma samples obtained from advanced ovarian carcinoma patients and healthy donors and corona-coated liposomes were subsequently recovered. Comprehensive comparison between ‘healthy’ and ‘diseased’ corona samples by label-free proteomics resulted in the identification of multiple differentially abundant proteins. Moreover, immunoassay-based validation of selected proteins demonstrated the potential of nanoparticle-platform proposed to discover novel molecules with great diagnostic potential. This study proposes a nanoparticle-enabled workflow for plasma proteomic analysis in healthy and diseased states and paves the way for further work needed to discover and validate panels of novel biomarkers for disease diagnosis and monitoring.
Keywords: Protein Corona, Biomarkers, Ovarian Cancer, Liposomes, Nanomedicine
Immunoassay
| FineTest Product | Sample | Detection Target | Species |
| Human PRKCSH(Glucosidase 2 subunit beta) ELISA Kit (EH2259) | Plasma | PRKCSH | Human |
As illustrated in Fig. 4B, ELISA quantification of the last two groups of proteins revealed higher performance than the clinically used biomarkers with AUC values between 97.6 % and 99.4%, while an AUC value of 100 % was shown for PDIA4 and PRKCSH proteins.
Validated Image
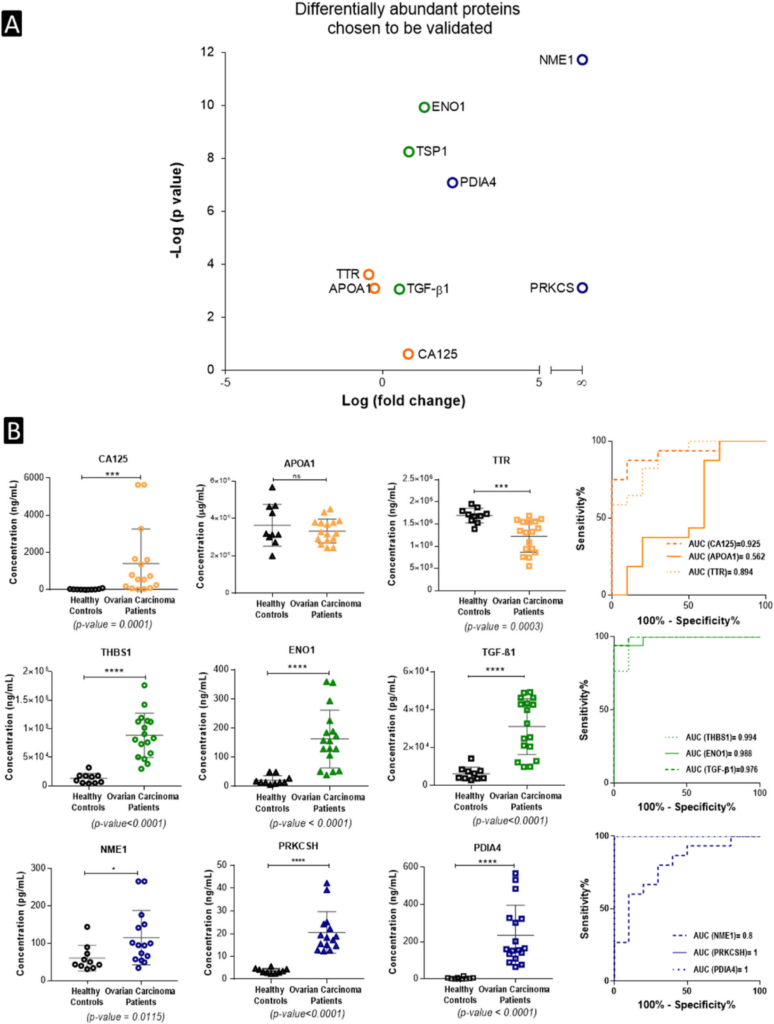
Figure Source: Nanotoday, 2020 Oct;34. doi: 10.1016/j.nantod.2020.100901.
Fig. 4. ELISA validation of the nanoparticle protein corona technology: (B) Plasma concentration profiles of selected potential biomarker proteins in healthy controls (n = 10) and ovarian carcinoma patients (n = 15-17) and their respective ROC curves based on ELISA assays. AUC values are also shown; Mann-Whitney t-test; * indicates p < 0.05, *** indicates p < 0.001, **** indicated p < 0.0001.
