FineTest ELISA kit contributes to the research on nanofiber wound dressing. The immunoassay is designed to measure lysozyme concentration in supernatant.
Publication Details
Article Title: Long-term antibacterial activity by synergistic release of biosafe lysozyme and chitosan from LBL-structured nanofibers
Journal Title: Carbohydrate Polymers
DOI: 10.1016/j.carbpol.2023.120791
IF: 10.723
PMID: 37059531
Abstract: Biosafe antibacterial agents are urgently demanded in treating infection especially chronic infection. However, efficient and controlled release of those agents remains great challenging. Two nature-derived agents, lysozyme (LY) and chitosan (CS), are selected to establish a facile method for long-term bacterial inhibition. We incorporated LY into the nanofibrous mats, then deposited CS and polydopamine (PDA) on the surface by layer-by-layer (LBL) self-assembly. In this vein, LY is gradually released with the degradation of nanofibers, and CS is rapidly disassociated from the nanofibrous mats to synergistically result in a potent inhibition against Staphylococcus aureus (S. aureus) and Escherichia coli (E. coli) over a period of 14 days. Besides long-term antibacterial capacity, LBL-structured mats could readily achieve a strong tensile stress of 6.7 MPa with an increase percentage of up to 103%. The enhanced proliferation of L929 cells arrives at 94% with help of CS and PDA on the surface of nanofibers. In this vein, our nanofiber has a variety of advantages including biocompatibility, strong long-term antibacterial effect, and skin adaptability, revealing the significant potential to be used as highly safe biomaterial for wound dressings.
Keywords: Chitosan, Layer-by-layer, Long-term antibacterial activity, Lysozyme, Nanofiber
Immunoassay
| FineTest Product | Sample | Detection Target | Species |
| Chicken LZM(Lysozyme LZM) ELISA Kit(ECH0086) | supernatant | Lysozyme | Chicken |
Illustration
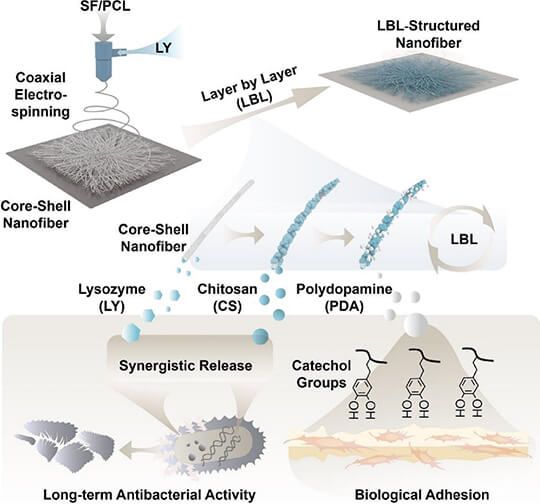
Figure Source: Carbohydr Polym. 2023 Jul 15;312:120791. doi: 10.1016/j.carbpol.2023.120791.
